 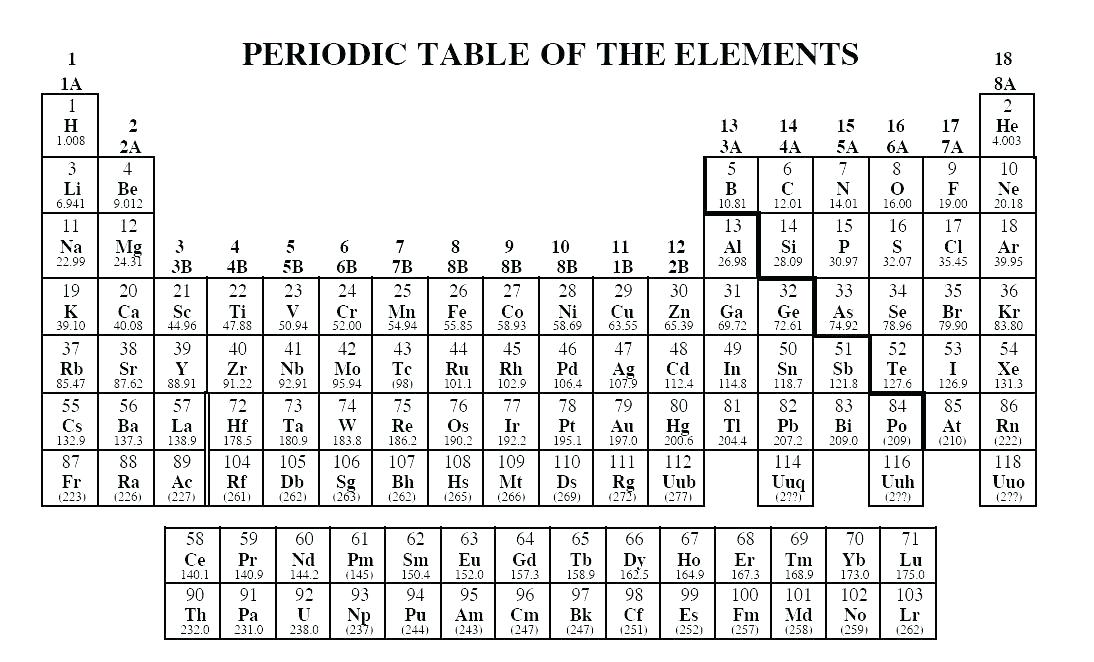
Go into any scientist’s office or lecture hall anywhere in the world and you are likely to see one. The metal was discovered and isolated in 1911 by Marie Curie.There is no more enduring reflection of science than the Periodic Table of Chemical Elements, which sheds light not only on the essence of chemistry but physics and biology as well. All isotopes of radium are radioactive and many exhibit luminescence, reacting readily with oxygen and water. Chemistry of Radium (Z=88) Radium takes its name from the Latin word radius or ray.Because of its reaction to air, barium cannot be found in nature in its pure form but can be extracted from the mineral barite. Chemistry of Barium (Z=56) Barium is a soft, silvery white metal, and has a melting point of 1000 K.Strontium is named after the Scottish village on Strontian. It has a silver appearance but then turns yellow with the formation of oxide. It is softer than calcium and decomposes water more vigorously. It occurs naturally only in compounds with other elements such as strontianite. Chemistry of Strontium (Z=38) Strontium is a group 2 element that does not occur as a free element due to its extreme reactivity with oxygen and water.All living organisms (in fact, even dead ones) have and need calcium for survival. Calcium is the fifth most abundant element by mass (3.4%) in both the Earth's crust and in seawater. It is a group 2 metal, also known as an alkaline-earth metal, and no populated d-orbital electrons. Chemistry of Calcium (Z=20) Calcium is the 20th element in the periodic table.Fine particles of magnesium can also catch on fire when exposed to air. Like aluminum, it forms a thin layer around itself to help prevent itself from rusting when exposed to air. Magnesium is light, silvery-white, and tough. Chemistry of Magnesium (Z=12) Magnesium is a group two element and is the eighth most common element in the earth's crust.Some Atypical Properties of Beryllium Compounds.It is not abundant in the environment and occurs mainly in the mineral beryl with aluminum and silicon. The element is a high-melting, silver-white metal which is the first member of the alkaline earth metals. Chemistry of Beryllium (Z=4) The name Beryllium comes from the Greek beryllos which is the name for the gemstone beryl.Sections below cover the trends in atomic radius, first ionization energy, electronegativity, and physical properties. Group 2: Physical Properties of Alkali Earth Metals This page explores the trends in some atomic and physical properties of the Group 2 elements: beryllium, magnesium, calcium, strontium and barium.The Thermal Stability of the Nitrates and Carbonates.The Solubility of the Hydroxides, Sulfates and Carbonates.Reactions of Group 2 Elements with Water.Reactions of Group 2 Elements with Oxygen.Reactions of Group 2 Elements with Acids.Includes trends in atomic and physical properties, trends in reactivity, the solubility patterns in the hydroxides and sulfates, trends in the thermal decomposition of the nitrates and carbonates, and some of the atypical properties of beryllium. Group 2: Chemical Properties of Alkali Earth Metals Covers the elements beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr) and barium (Ba). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
